A supplement cocktail that lowers glycation stress can partly rewind hormone driven liver aging, a finding that reshapes how scientists think about growth hormone and organ decline.
In a study published October 3 2025 in Aging-US, researchers from the Buck Institute for Research on Aging, Southern Illinois University School of Medicine, and the University of Arizona used bovine growth hormone transgenic mice to show that chronic growth hormone excess disrupts metabolism, activates inflammation, and pushes the liver into an aging like state, while a glycation lowering mix called Gly-Low reverses many of these shifts.
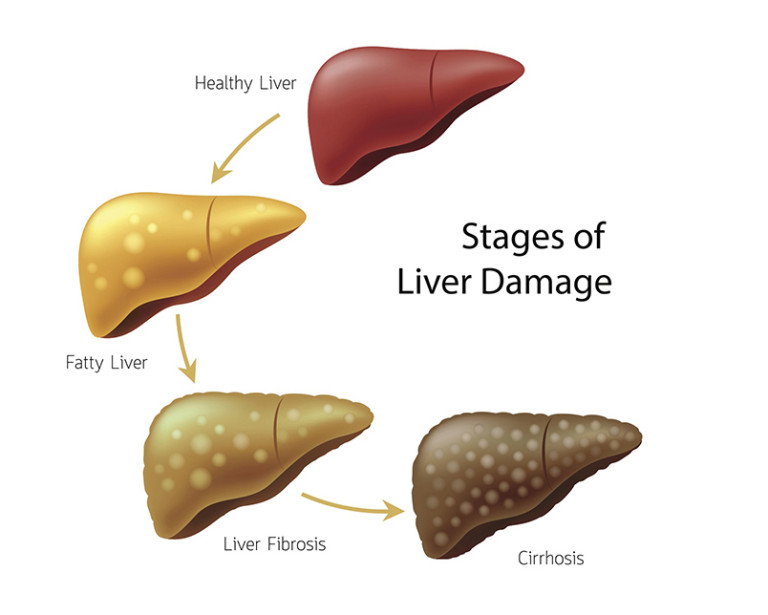
Growth hormone is essential for development, but when levels remain chronically elevated, as in acromegaly or some pituitary tumors, organ systems can falter. In the liver, the team found, that disruption takes the form of oxidative stress, suppressed lipid metabolism, and immune activation. Under the microscope, livers from growth hormone overloaded mice showed the kind of patchy, swollen tissue and immune cell crowding usually seen in much older animals, a visual signal of how fast the hormone pushes biology off balance.
Hormone Excess Mimics Decades of Liver Aging
Using bulk RNA sequencing on liver tissue from young bGH-Tg mice, the team mapped more than five thousand gene expression changes. Metabolic pathways that normally handle fat oxidation, bile secretion, and detoxification were dialed down. Immune and inflammatory pathways, including cytokine signaling and leukocyte activation, were turned up. It was a molecular profile that looked less like youth and more like late life.
Remarkably, the liver transcriptomic profile of young bGH-Tg mice mirrors that of the aged liver, with pronounced signs of cellular senescence and AGE accumulation.
That overlap was not subtle. When the team compared growth hormone induced signatures to normal aging signatures, they found 596 genes changing in both datasets with a significant positive correlation. The liver, in effect, was aging early. Advanced glycation end products sat at the center of that shift. Mass spectrometry revealed elevated levels of AGEs such as CML and MGH 1, paired with lower glutathione redox ratios and reduced NADPH, markers of oxidative strain.
Key regulatory proteins reflected the same pattern. SREBF1, CLOCK, and PPAR alpha, which help maintain lipid balance and metabolic rhythm, were reduced in bGH-Tg livers. Pathway analyses pointed toward immune regulators such as NF kappa B and STAT1 as drivers of the inflammatory tilt. It was the kind of biochemical constellation that often accompanies liver decline in old age, but here it was appearing in months, not years.
A Glycation Lowering Mix Rewinds Molecular Damage
Here is where the story pivots. When the team added Gly-Low, a cocktail of nicotinamide, alpha lipoic acid, thiamine, piperine, and pyridoxamine, to the animals’ diets for up to 40 weeks, the trajectory changed. AGE levels dropped. Insulin sensitivity improved. Fasting glucose, glucose tolerance, and fat mass all shifted toward healthier ranges. Mice on Gly-Low ran longer on the rotarod and gripped harder in strength tests, behavioral hints of restored metabolic stability.
The liver’s internal wiring also began to reset. The researchers identified 235 differentially expressed genes between treated and untreated bGH-Tg mice, with 163 overlapping the growth hormone induced signature. Crucially, these shared genes moved in opposite directions, a strong negative correlation that signaled reversal rather than compensation.
“Glycation-lowering strategies may serve as effective treatments for alleviating GH-induced metabolic and inflammatory disruptions in the liver, offering a promising avenue for addressing age-related metabolic diseases associated with GH dysregulation.”
Pathways regained footing. Genes involved in oxidoreductase activity, estrogen metabolism, adenylate cyclase regulation, and PPAR signaling rose back toward normal. Genes linked to cAMP signaling, gluconeogenesis, and inflammatory mediator regulation fell. PPARA, SREBF1, ATF6, CLOCK, and HNF1A emerged again as central nodes through which the reversal unfolded. Even at the protein level, PPAR alpha expression, diminished in bGH-Tg mice, was restored with treatment.
The findings do not claim that Gly-Low cures hormone driven aging, but they show that a major component of the damage, glycation stress, is modifiable. In a field where aging processes often feel irreversible, the idea that a hormone accelerated decline can be partly unwound is both biologically compelling and therapeutically notable.
Aging-US: 10.18632/aging.206327
If our reporting has informed or inspired you, please consider making a donation. Every contribution, no matter the size, empowers us to continue delivering accurate, engaging, and trustworthy science and medical news. Independent journalism requires time, effort, and resources—your support ensures we can keep uncovering the stories that matter most to you.
Join us in making knowledge accessible and impactful. Thank you for standing with us!